COVID-19 Testing
Seaport Diagnostics is a leading provider of COVID-19 testing. Our national scale and comprehensive clinical and digital solutions enable us to provide a seamless collection, testing and reporting solution.
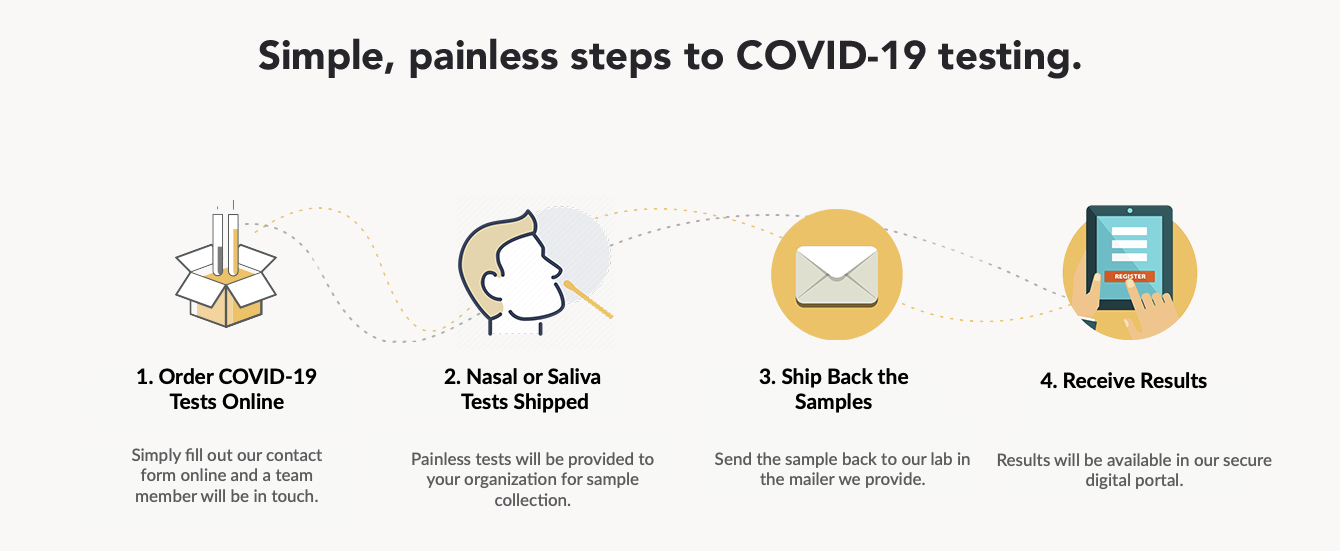
Seaport Diagnostics offers the following services:
- Weekly and Bi-Weekly Testing programs available
- Nasal swab and saliva testing options
- Results with 24/36 hours of receipt at lab
- Programs for returning workers
- Executive traveller programs available
- Onsite collection available in certain areas
Seaport Diagnostics is able to directly bill insurance instead of your organization for reimbursable COVID-19 tests. Our testing solutions start at $100. Contact us to learn more!
Coronavirus (COVID-19) Information
 What is coronavirus disease (COVID-19)?
What is coronavirus disease (COVID-19)?- Coronavirus disease (also called COVID-19) is an infection caused by SARS-CoV-2 (severe acute respiratory syndrome coronavirus), one of the most recently discovered types of coronaviruses. Those who have this disease may or may not experience symptoms, which range from mild to severe.
 How does COVID-19 spread?
How does COVID-19 spread?- COVID-19 spreads from person-to-person. When an infected person coughs, sneezes, or exhales air, droplets containing the virus go into the air and onto surfaces and objects around them. Other people are exposed to the virus by breathing in these droplets or by touching their eyes, nose, or mouth after touching infected surfaces.
 What are the symptoms of COVID-19?
What are the symptoms of COVID-19?Symptoms may appear 2 to 14 days after being exposed to the virus. The most common symptoms include:
- Fever
- Cough
- Shortness of breath or difficulty breathing
- Chills
- Repeated shaking with chills
- Muscle pain
- Headache
- Sore throat
- New loss of taste or smellCases of COVID-19 range from mild to severe. Some people who are infected don’t have any symptoms and don’t feel sick. Most people have mild symptoms.
 Who is at high risk of getting very sick?
Who is at high risk of getting very sick?- Severe cases are more likely to occur in older adults (65 years of age and older), as well as pregnant women, those with weakened immune systems, and those with underlying health issues (such as lung disease, diabetes, obesity, high blood pressure, heart conditions, stroke, kidney disease or on dialysis, liver disease, cancer, transplant, AIDS, lupus, and rheumatoid arthritis). However, serious illness can also occur in young, healthy adults.
 How is COVID-19 treated?
How is COVID-19 treated?- There is currently no treatment for COVID-19. Not all patients with COVID-19 will require medical attention, and most people recover within 2 weeks without any specific treatment. For severe cases, hospitalization and respiratory support may be required. For mild cases, treatment focuses on managing symptoms.
 What is a COVID-19 PCR test?
What is a COVID-19 PCR test?- PCR (polymerase chain reaction) tests check for genetic material (viral RNA) produced by the virus. This determines if an individual is actively infected with COVID-19 and can spread it to others.
 Who should get a COVID-19 PCR test?
Who should get a COVID-19 PCR test?-
You should get tested if:
- You have symptoms of COVID-19, are a healthcare worker, or you live or work in a place where people reside, meet, or gather in close proximity. This can include homeless shelters, assisted living facilities, group homes, prisons, detention centers, schools, and workplaces.
- A public health department or contact investigator has identified you as someone who should get tested.
This test may also be helpful if:
- You have symptoms of COVID-19 and want to check to see if you have the virus and can pass it on to others.
- You do not have symptoms but the health department or your clinician identified you as someone who should get tested.
If you are a healthcare professional, first responder, frontline worker, or critical infrastructure worker and believe you have been directly exposed while at work, you should consult your place of work for specific occupational health guidance about testing and whether to stay home or continue working while waiting for your results. You should adhere to recommendations set forth by your employer or your local department of health, as they may differ from the CDC’s guidelines.
If you have any other questions, we recommend that you speak with your primary healthcare provider about testing recommendations to see if testing is right for you at this time.  What will my COVID-19 PCR test results tell me?
What will my COVID-19 PCR test results tell me?- Your test results will confirm whether or not you are currently infected with COVID-19. If your results are positive, it is important to limit your exposure to other people and continue to monitor your symptoms. If your results are negative, the virus was not present in the sample you provided.
 Are there any limitations to COVID-19 PCR tests?
Are there any limitations to COVID-19 PCR tests?- This test may not detect the virus in early stages of infection. Furthermore, the virus may not be detected if the sample is not collected as directed. Although the possibility is low, a false negative result should be considered if you have had recent exposure to the virus along with symptoms consistent with COVID-19.
 How do I prepare for the test?
How do I prepare for the test?- You do not need to do anything to prepare for the test. You do not need to fast or stop taking any medications before testing. Further instructions will be provided to you in your test kit.
 How do I collect my COVID-19 test sample?
How do I collect my COVID-19 test sample?- We offer saliva and nasal tests. Specific instructions on how to conduct the test(s) will be included with your order. For our anterior nasal tests, Please see the instructions from the CDC at the following link: https://www.cdc.gov/coronavirus/2019-ncov/downloads/community/COVID-19-anterior-self-swab-testing-center.pdf.
The Seaport Diagnostics SARS-CoV-2/COVID-19 Test is designed to detect the presence of the underlying virus (SARS-CoV-2) that causes COVID-19, and is for use with patients who meet current guidance for evaluation. This test has not been FDA cleared or approved.
The SARS-CoV-2 RNA is generally detectable in respiratory specimens during the acute phase of infection. Positive results are indicative of the presence of SARS-CoV-2 RNA; clinical correlation with patient history and other diagnostic information is necessary to determine patient infection status. Positive results do not rule out bacterial infection or co-infection with other viruses. The agent detected may not be the definite cause of disease. Negative results do not preclude SARS-CoV-2 infection and should not be used as the sole basis for patient treatment or other patient management decisions. Negative results must be combined with clinical observations, patient history, and epidemiological information.
Laboratories within the United States and its territories are required to report all positive (and in many cases, negative) results to the appropriate public health authorities.